How heat pumps work
In order to understand how heat pumps work one must first understand what heat is. Many people confuse heat with temperature and thus have trouble understanding how it is possible to pump heat from outside where the temperature may be a cold 8 degrees and yet somehow get the temperature inside the house to a pleasant 22 degrees.
Heat is a form of energy just like light is a form of energy. The amount of heat in a substance is partly related to its temperature but also to its physical properties and its volume. To give an example lets consider two common day substances, air and water, and a common container such as a jar. If one jar was filled with water heated to 25 degrees, and another identical jar just contained air at 25 degrees, they would not contain the same amount of heat even though they are at the same temperature. The jar of water would contain substantially more heat. If you placed both jars in the fridge it would take a lot longer for the jar of water to get cold. The hotter something is the more heat it will contain but how much heat it contains at a given temperature is related to what the substance is and its volume. A big jar of water at 25 degrees will contain more heat than a small jar of water at 25 degrees. A small jar of water at 25 degrees will contain more heat than a big jar of air at 25 degrees.
Once you have got an understanding of what heat is then you need to know some fundamental principles of heat. The first principle is that heat will always flow from hot to cold and try to reach an equilibrium. That is why it is impossible for you to have a jar that has half of its water hot and the other half cold. The heat will quickly flow from the hot half to the cold half until all the water becomes the same temperature more or less. I say more or less because there is another physical principle whereby hot water is lighter than cold water and hence will rise to the surface. However in the example I mentioned with the jar you would not notice this effect.
Another principle is that if you compress a gas or a mixture of gases such as air, it will get hot. That is why a bicycle pump gets hot when you pump up a tyre. Each time you push the pump the air compresses and gets warmer.
Most people are familiar with the previous principle but what is less well known is that the opposite also happens. When a gas expands it gets colder. If you take an aerosol can of air freshener you may notice that as the highly compressed gas escapes from its can it expands and becomes cooler.
One other important principle is that if sufficient heat is applied to a liquid it will change into a gas. There will be a maximum temperature for any liquid known as its boiling point where no matter how much you heat the liquid it will remain at this temperature while changing state from a liquid to a gas. This changing of state is known as a phase change. Likewise, for any gas there is a temperature where it will condense into a liquid and heat will be released. A liquid to gas phase change is an endothermic change as it requires the input of heat. A gas to liquid phase change is an exothermic change as it releases heat. Every liquid has a different boiling point. For water this temperature is 100 degrees. For refrigerant liquids this temperature is considerably lower.
Lastly before we explain how a heat pump works we must realise that no matter how cold the air is it still contains heat, even at an unbearably cold -45 degrees it still contains heat.
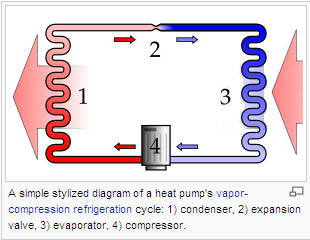
The magic of a heat pump is that it can do the 'apparent' reverse of the first principle I mentioned. It can make heat flow from a cold place to a hot place. It does this magic by exploiting the other principles I mentioned. A heat pump has 2 components, an outside component and an inside component connected by a loop of pipe filled with a special type of gas called a refrigerant. When you want the heat pump to warm your home the outside unit will make the refrigerant change phase from liquid to gas and in the process suck in heat from the outside air. The heat pump will then move this gas to the indoor unit where the gas gets compressed and forced to condense thereby releasing heat to the surrounding air. The refrigerant then returns to the outdoor unit where the cycle is repeated.
A heat pump can pump heat in either direction thereby warming or cooling a room. A refrigerator is none other than a heat pump that pumps heat out of your fridge and into your kitchen. If you touch the back of your fridge you will notice it is hot.